Paper of the Month - March 2026
selected by the BMAS Scientific Board
A catecholamine-independent pathway controlling adaptive adipocyte lipolysis.
- 1Division of Bone and Mineral Diseases, Washington University School of Medicine, St. Louis, MO, USA.
- 2Department of Biomedical Engineering, Washington University in St. Louis, St. Louis, MO, USA.
- 3Department of Developmental Biology, Washington University in St. Louis, St. Louis, MO, USA.
- 4Division of Endocrinology, Metabolism and Lipid Research, Washington University School of Medicine, St. Louis, MO, USA.
- 5Department of Neuroscience and Department of Psychiatry, Washington University in St. Louis, St. Louis, MO, USA.
- 6Department of Biochemistry and Molecular Biology, Mayo Clinic, Rochester, MN, USA.
- 7Department of Molecular and Integrative Physiology, University of Michigan, Ann Arbor, MI, USA.
- 8Program in Physical Therapy and Departments of Neurology and Orthopaedic Surgery, Washington University School of Medicine, St. Louis, MO, USA.
- 9Systems Pharmacology and Translational Therapeutics, University of Pennsylvania, Philadelphia, PA, USA.
- 10Division of Bone and Mineral Diseases, Washington University School of Medicine, St. Louis, MO, USA. scheller@wustl.edu.
- 11Department of Biomedical Engineering, Washington University in St. Louis, St. Louis, MO, USA. scheller@wustl.edu.
- 12Department of Developmental Biology, Washington University in St. Louis, St. Louis, MO, USA. scheller@wustl.edu.
- #Contributed equally.
Nature Metabolism. 2026 Jan;8(1):96-115.
PMID: 41507664 | PMCID: PMC12855016 | DOI: 10.1038/s42255-025-01424-5
Correspondence:Erica L Scheller scheller@wustl.edu.
Paper Highlight
Constitutive bone marrow adipose tissue (cBMAT) is largely resistant to conventional lipolytic stimuli, including acute fasting, caloric restriction, exercise, β-adrenergic agonists, and cold exposure. However, cBMAT can be depleted under extreme metabolic stress, such as severe anorexia, late stages of starvation, or pathological conditions associated with severe wasting and cachexia.
In a recent study, Zhang et al. identified a novel mouse model in which chronic (9-day) intracerebroventricular (ICV) leptin infusion mimics the end-stage depletion of BMAT. Surprisingly, this catabolic process appears to be independent of local peripheral nerves, sympathetic nervous system (SNS) signaling, or catecholamines. Instead, cBMAT depletion is associated with systemic hypoinsulinemia and hypoglycemia and is mediated by adipose triglyceride lipase (ATGL)–dependent lipolysis. Furthermore, the balance between the lipolytic inhibitor G0S2 and ATGL appears to regulate the lipolytic resistance of cBMAT.
Key Findings
- ICV leptin infusion rapidly depletes cBMAT independent of reduced food intake.
- ICV leptin–induced cBMAT depletion is not mediated by peripheral nerves, the SNS, or catecholamines.
- Hypoinsulinemia and hypoglycemia contribute to cBMAT catabolism following ICV leptin treatment.
- ICV leptin activates lipolysis in an ATGL-dependent manner.
- The ratio of the lipolytic inhibitor G0S2 to ATGL is associated with the lipolytic resistance of cBMAT under baseline conditions, during aging, and after ICV leptin treatment.
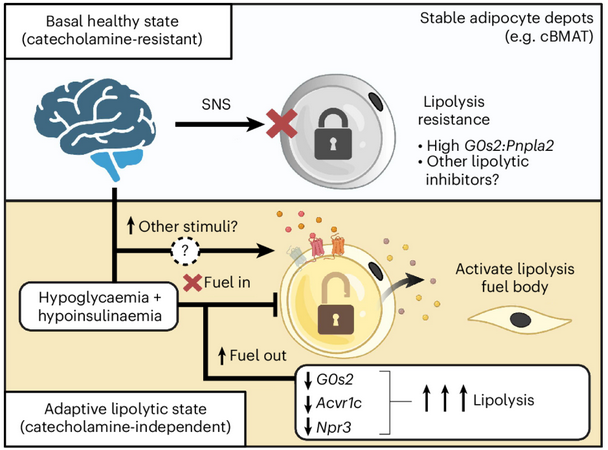
Figure: Summary of Findings with ICV leptin translate to a model of tumour-evoked cachexia.(Zhang et al, Figure 8i) © The Authors 2026
