Paper of the Month - June 2025
selected by the BMAS Scientific Board
Uncovering mechanisms of thiazolidinediones on osteogenesis and adipogenesis using spatial fluxomics
Kristyna Brejchova1, Michal Rahm2, Andrea Benova3, Veronika Domanska1, Paul Reyes-Gutierez2, Martina Dzubanova4, Radka Trubacova1, Michaela Vondrackova1, Tomas Cajka5, Michaela Tencerova3, Milan Vrabel2, Ondrej Kuda6
1 Laboratory of Metabolism of Bioactive Lipids, Institute of Physiology of the Czech Academy of Sciences, Videnska 1083, 14200 Prague, Czechia.
2 Chemistry of Bioconjugates, Institute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences, Flemingovo nám. 2, 166 10 Prague, Czechia.
3 Laboratory of Molecular Physiology of Bone, Institute of Physiology of the Czech Academy of Sciences, Videnska 1083, 14200 Prague, Czechia.
4 Laboratory of Molecular Physiology of Bone, Institute of Physiology of the Czech Academy of Sciences, Videnska 1083, 14200 Prague, Czechia; Faculty of Science, Charles University, Albertov 6, 128 00 Prague, Czech Republic.
5 Laboratory of Translational Metabolism, Institute of Physiology of the Czech Academy of Sciences, Videnska 1083, 14200 Prague, Czechia.
6 Laboratory of Metabolism of Bioactive Lipids, Institute of Physiology of the Czech Academy of Sciences, Videnska 1083, 14200 Prague, Czechia.
Correspondence: ondrej.kuda@fgu.cas.cz.
Abstract
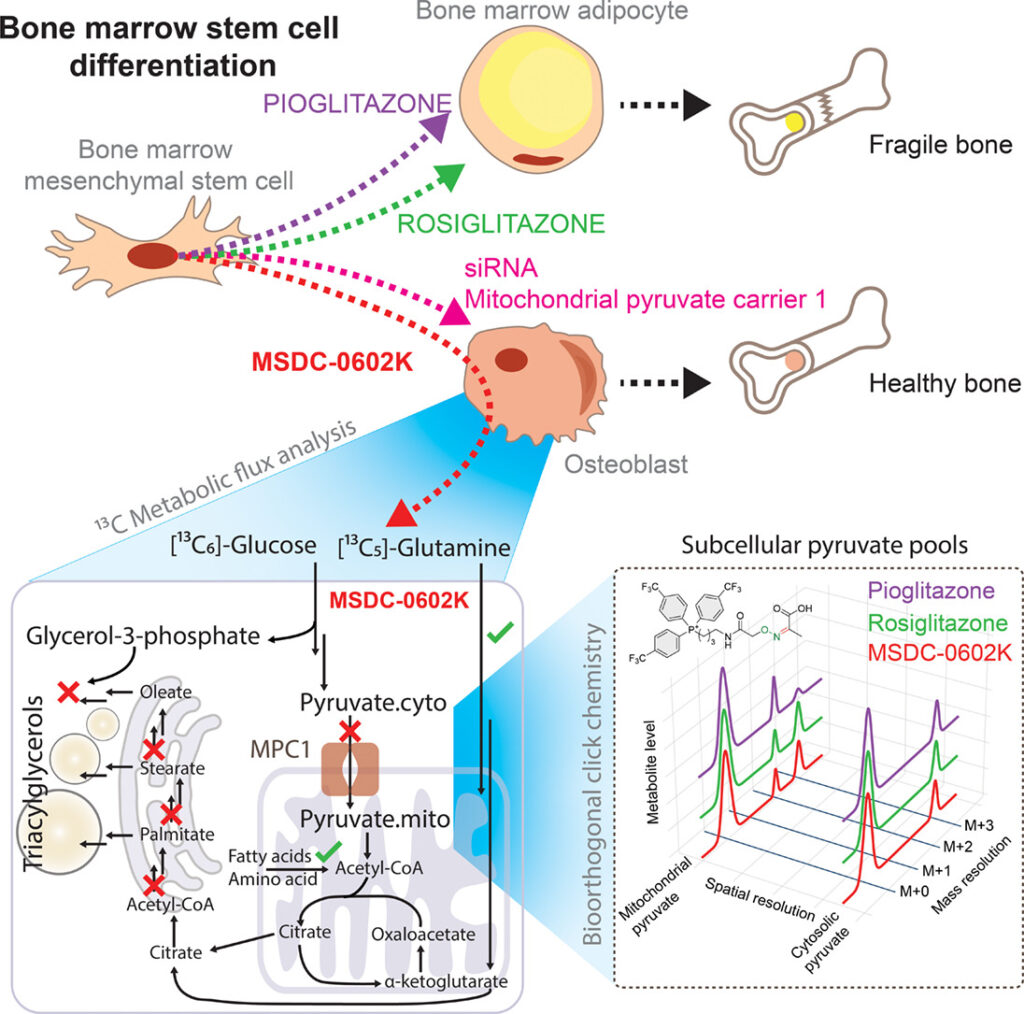
Objective: Insulin-sensitizing drugs, despite their broad use against type 2 diabetes, can adversely affect bone health, and the mechanisms underlying these side effects remain largely unclear. Here, we investigated the different metabolic effects of a series of thiazolidinediones, including rosiglitazone, pioglitazone, and the second-generation compound MSDC-0602K, on human mesenchymal stem cells (MSCs).
Methods: We developed 13C subcellular metabolomic tracer analysis measuring separate mitochondrial and cytosolic metabolite pools, lipidomic network-based isotopologue models, and bioorthogonal click chemistry, to demonstrate that MSDC-0602K differentially affected bone marrow-derived MSCs (BM-MSCs) and adipose tissue-derived MSCs (AT-MSCs). In BM-MSCs, MSDC-0602K promoted osteoblastic differentiation and suppressed adipogenesis. This effect was clearly distinct from that of the earlier drugs and that on AT-MSCs.
Results: Fluxomic data reveal unexpected differences between this drug’s effect on MSCs and provide mechanistic insight into the pharmacologic inhibition of mitochondrial pyruvate carrier 1 (MPC). Our study demonstrates that MSDC-0602K retains the capacity to inhibit MPC, akin to rosiglitazone but unlike pioglitazone, enabling the utilization of alternative metabolic pathways. Notably, MSDC-0602K exhibits a limited lipogenic potential compared to both rosiglitazone and pioglitazone, each of which employs a distinct lipogenic strategy.
Conclusions: These findings indicate that the new-generation drugs do not compromise bone structure, offering a safer alternative for treating insulin resistance. Moreover, these results highlight the ability of cell compartment-specific metabolite labeling by click reactions and tracer metabolomics analysis of complex lipids to discover molecular mechanisms within the intersection of carbohydrate and lipid metabolism.
Graphical Abstract
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
